Musculoskeletal Regenerative Engineering
The Guldberg lab is leveraging advanced biofabrication techniques like 6-axis bioprinting (above), microfluidics (below, left and center) and selective laser melting (below, right) to engineer the next generation of medical devices and in vitro organoids.
-
Microfluidics Design
Dr. Mancipe-Castro holds a microfluidics chip used to produce micron-scale cross-linked hydrogels. Modified to bind to and encapsulate joint cells, they can serve both as cell and drug delivery vehicles and as scalable organoids to study osteoarthritis.

-
Microgel Synthesis
Video of poly(ethylene glycol) (PEG) polymer being crosslinked in a microfluidics chip to form microgels. Taken under 10X objective microscope.

-
3D-Printed Titanium Scaffolds
3D micro-CT reconstruction of Titanium implant produced by selective laser melting. Guldberg lab is collaborating with company Restor3D to study these Ti bone implants with unique gyroid architecture as osteoinductive scaffolds for bone repair.
Developing therapeutics and monitoring disease progression in osteoarthritis
Osteoarthritis (OA) affects nearly 21 million people in the United States and is one of the leading causes of chronic disability.
Osteoarthritis disease progression leads to degradation of articular cartilage and subchondral bone as well as changes to the synovium, blood vessels, tendons, and muscle that cause pain and decrease mobility. Currently, only symptom management treatments are available to the patient population. One key research area for the Guldberg lab is to develop and evaluate disease modifying osteoarthritis drugs (DMOADs) and therapeutics. We approach this goal by developing scalable organoid in vitro models and pre-clinical models which can be used as testbeds for novel drugs, cellular therapies and biomaterials. We focus both on engineering new therapeutics and identifying new biomarkers to diagnose and track disease progression. We are also interested in applying regenerative rehabilitation principles to the treatment of OA, combining cellular and drug delivery therapies with customized rehabilitation regimens.
Our approach to osteoarthritis disease characterization, in vitro modeling, and therapy.
Harnessing mechanical regulation of musculoskeletal regeneration
Regenerative rehabilitation takes advantage of mechanically regulated biological processes to enhance tissue repair.
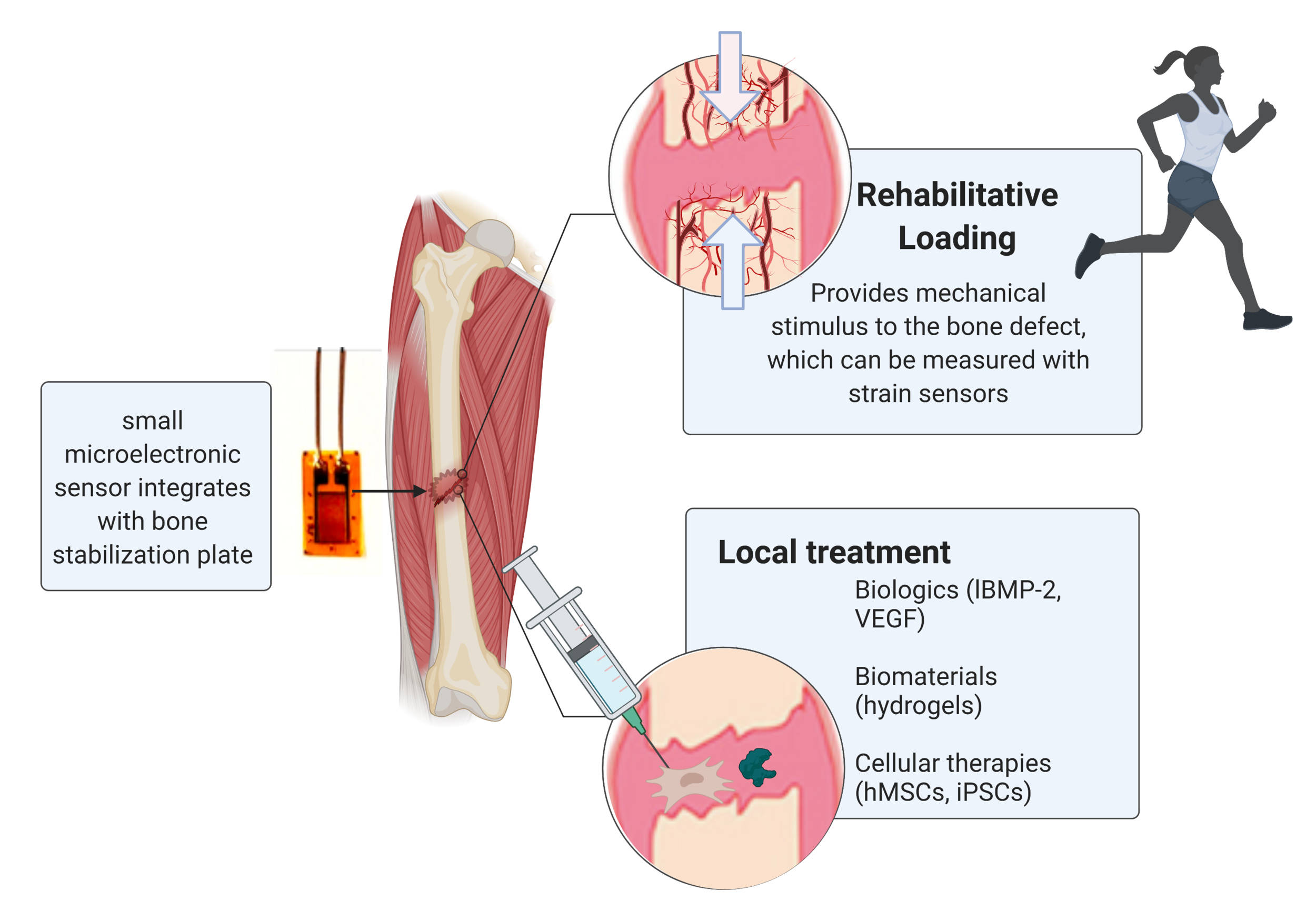
Regenerative Rehabilitation combines mechanical stimulation with local therapy.
A crucial research area for the Guldberg lab is Regenerative Rehabilitation, a multidisciplinary field which takes a two-pronged approach to healing traumatic injury: mechanical stimulation via rehabilitative loading and local treatments like biologics and biomaterials. As such, our goal is to improve our understanding of the complex mechanical environment at an injury site and its influence on the local and systemic cellular processes. Advances in micro-electronic systems (MEMS) have allowed us to create small implantable sensors that permit real-time analysis of in vivo mechanical environments during musculoskeletal healing. The implanted strain sensor combined with a transceiver wirelessly transmit quantitative measurements of the local mechanical environment during regeneration. When implanted in conjunction with therapies or tissue engineered constructs, strain sensors enable an advanced understanding of mechanobiology throughout the regenerative process, thus providing greater insight into the effectiveness of and mechanisms behind potential regenerative therapies.
Another goal is to understand temporal-mechanical interplay and its effects on regeneration. We use both pre-clinical and in vitro models to assess vascular and bone growth under different load conditions, considering both acute and delayed treatment models. This work is relevant to designing advanced patient-specific medical devices and rehabilitation protocols.
Engineering the body’s immune response to promote healing after trauma
Minimally invasive blood draws may allow us to detect and treat immune dysregulation.
The role of local and systemic immune responses in musculoskeletal trauma.
Clinically, traumatic musculoskeletal injury can result in prolonged dysregulation of the immune system, which can lead to poor healing outcomes. In particular, cells known as Myeloid Derived Suppressor Cells (MDSCs) can be detected in elevated levels after trauma and have been correlated with reduced bone formation. MDSCs can increase the proliferation of T regulatory cells and suppress cytotoxic T and natural killer cells, contributing to chronic suppression of the inflammatory response. Our goal is to prevent immune dysregulation by targeting and depleting problematic immune suppressor cells. Engineered synthetic nanoparticle antibodies (SNAbs) (right) colocalize the problematic MDSCs with activated macrophages, resulting in their depletion. We are currently studying whether MDSC depletion concurrent with local growth factor delivery can improve bone healing.
Synthetic Nanoparticle Antibody (SNAb)
STEM-BF, STEM-HAADF, and EDS mapping image of a SNAb, a Janus (two-sided) functionalized gold nanoparticle. Biotinylated cadmium quantum dots bind to streptavidin coating on one side of the Janus nanoparticle. Adapted from Liu, et al, Nano Letters, 2021.